In Arabidopsis, two copies of IRE1, Arabidopsis thaliana (At)IRE1a and AtIRE1b, and two copies of the transmembrane bZIP, AtbZIP28, and AtbZIP17, with partially overlapping functions operate in UPR. Plant UPR is transduced as a well-characterized bipartite signaling module consisting of the ER membrane-associated transducers inositol-requiring protein 1 (IRE1) and bZIP (basic leucine zipper) transmembrane transactivation factors. 
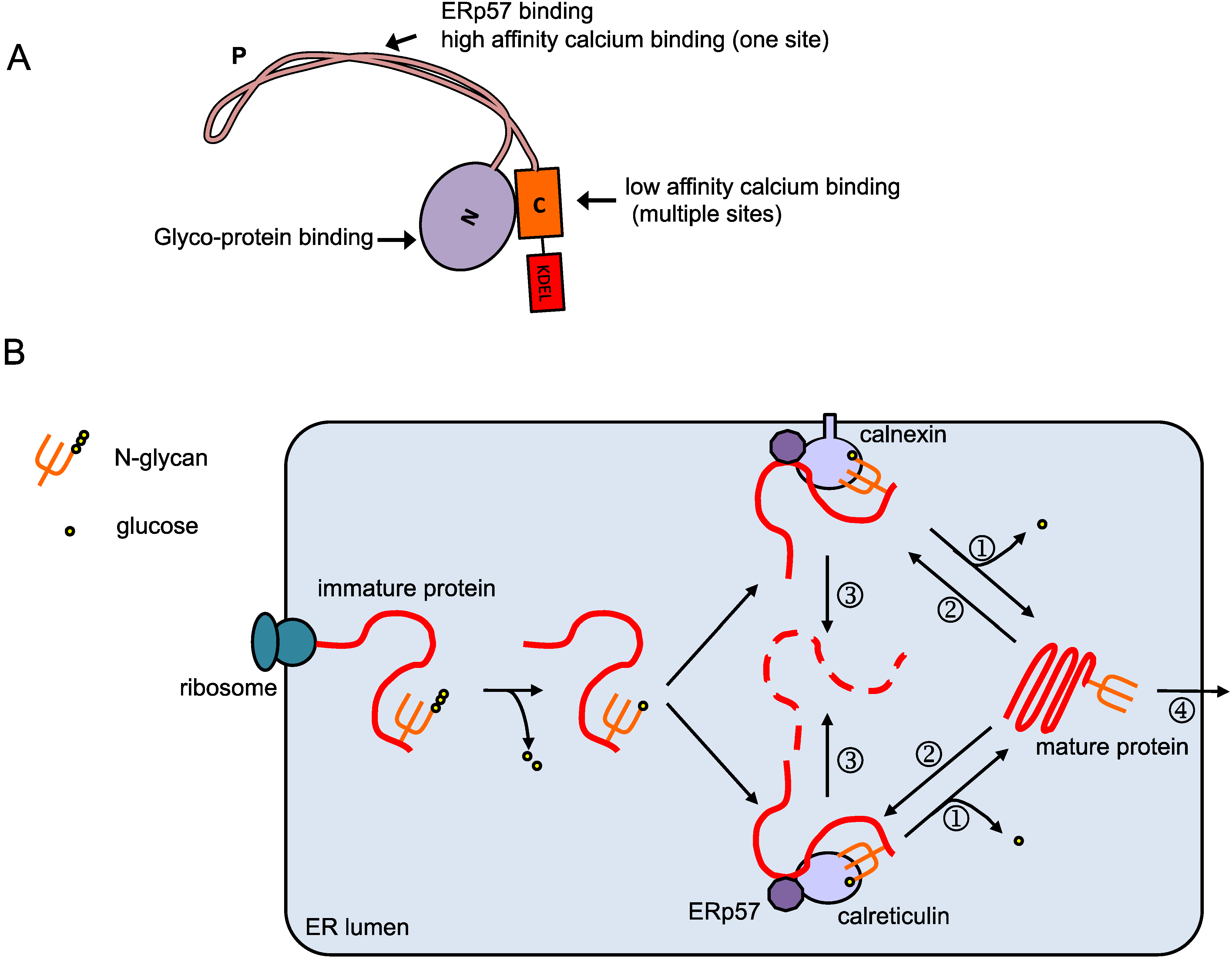
The unfolded protein response (UPR) is an evolutionarily conserved signaling pathway activated in response to ER stress. The perturbations in the ER function activate signaling cascades that allow ER communication with the cytoplasm, nucleus, and, under chronically prolonged ER stress, mitochondria, and vacuole to restore ER homeostasis or ultimately cause programmed cell death (PCD). It addresses misfolded proteins to degradation via either the ER-associated degradation (ERAD) system or autophagy. To minimize the deleterious effect of misfolded proteins and prevent their translocation further in the secretory pathway, a protein quality control machinery monitors protein folding. Stress conditions that disturb this equilibrium and promote the accumulation of unprocessed, misfolded protein in the organelle promote ER disfunction, a process known as ER stress. Under normal conditions, the rate of protein processing in the ER lumen is balanced with the protein synthesis rate and loading into the organelle. These protein processing activities allow nascent proteins to their destination in the secretory pathway. It is a multitask intracellular organelle that provides the functional apparatus for translocation of the newly synthesized secretory proteins to the lumen of the organelle, protein folding, and protein post-translational modifications. The endoplasmic reticulum (ER) is the gateway of synthesized proteins by ER membrane-bound polysomes to the secretory pathway. We also highlight the conserved features of ER stress-induced cell death signaling in plants shared by eukaryotic cells. Here, we summarize recent advances in understanding plant-specific molecular mechanisms that elicit cell death signaling from ER stress. Accumulating evidence implicates ER stress-induced cell death signaling pathways as significant contributors for stress adaptation in plants, making modulators of ER stress pathways potentially attractive targets for stress tolerance engineering. However, if the ER stress persists and cannot be reversed, the chronically prolonged stress leads to cellular dysfunction that activates cell death signaling as an ultimate attempt to survive. The recovery from ER stress is accomplished by decreasing protein translation and loading into the organelle, increasing the ER protein processing capacity and ER-associated protein degradation activity. In eukaryotic cells, the evolutionarily conserved unfolded protein response is activated to clear unfolded proteins and restore ER homeostasis. The endoplasmic reticulum (ER) stress response is triggered by any condition that disrupts protein folding and promotes the accumulation of unfolded proteins in the lumen of the organelle. Department of Biochemistry and Molecular Biology, BIOAGRO, National Institute of Science and Technology in Plant-Pest Interactions, Universidade Federal de Viçosa, Viçosa, Brazil.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
